1.Product Overview
| Product Name |
Valganciclovir Tablets, USP |
| Reference Listed Drug (RLD) |
Xarelto®(Janssen) |
| Pharmaceutical Form |
Film-coated tablet |
| Strength |
450 mg |
2.Indications and Usage
Valganciclovir is a prodrug of ganciclovir that is indicated for:
| Adult Patients |
Treatment of CMV Retinitis |
In patients with Acquired Immunodeficiency Syndrome (AIDS) |
| Prevention of CMV Disease |
In kidney, heart, or kidney-pancreas transplant patients at high risk (Donor CMV seropositive/Recipient CMV seronegative [D+/R-]) |
| Pediatric Patients |
Prevention of CMV Disease: In kidney transplant patients (4 months to 16 years) and heart transplant patients (1 month to 16 years) at high risk |
3.Dosage and Administration
| Important Administration Instructions Caution |
Valganciclovir tablets should be taken with food. |
| Valganciclovir tablets are a prodrug of ganciclovir; oral valganciclovir 900 mg provides systemic ganciclovir exposure comparable to IV ganciclovir 5 mg/kg. |
| Dosage must be adjusted according to patient's renal function (Creatinine Clearance, CrCl). |
Standard Adult Dosing:
| Indication |
Dosage |
Duration |
| CMV Retinitis (Treatment) |
Induction: 900 mg (two 450 mg tablets) twice daily |
21 days |
| Maintenance: 900 mg (two 450 mg tablets) once daily |
- |
| CMV Disease (Prevention) |
900 mg (two 450 mg tablets) once daily |
Start within 10 days of transplant, continue until 100 days post-transplant. |
4.Comparison of Oral Antivirals for CMV Management
| Feature |
Valganciclovir (Generic) |
Ganciclovir (Oral) |
Letermovir |
| Mechanism of Action (MOA) |
Prodrug of Ganciclovir. Inhibits viral DNA synthesis by competing with deoxyguanosine triphosphate. |
Ganciclovir. Inhibits viral DNA synthesis. (Same active moiety as Valganciclovir.) |
CMV Terminase Complex Inhibitor. Inhibits viral DNA processing and packaging. |
| Primary Oral Indication(s) |
Treatment of CMV retinitis. Prevention of CMV disease in solid organ transplant (SOT). |
Prevention of CMV disease in transplant recipients. Maintenance for CMV retinitis (less common). |
Prevention of CMV infection and disease in adult CMV-seropositive recipients of an allogeneic hematopoietic stem cell transplant (HSCT). |
| Bioavailability (Oral) |
High (~60%) |
Very Low (~6–9%) |
Moderate (Varies with/without cyclosporine). |
| Standard Adult Dosing |
900 mg once or twice daily (depending on indication). |
1000 mg three times daily. |
480 mg once daily. |
| Key Differentiator |
High bioavailability allows for once/twice-daily dosing, replacing IV ganciclovir. |
Low bioavailability requires frequent dosing (TID) and large pill burden. |
Novel MOA; specifically for CMV prophylaxis in the HSCT setting. |
5.Advantages of Our Generic Valganciclovir Tablets
| Cost-Effectiveness |
Provides a highly effective antiviral therapy at a more accessible price point compared to the brand-name drug, reducing the financial burden on patients and payers. |
| Therapeutic Equivalence (A-Rated) |
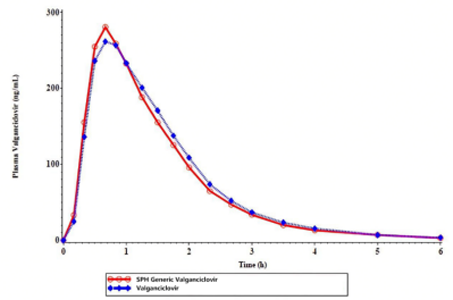
As an FDA-approved generic (ANDA 218027), it is bioequivalent to the reference drug (Valcyte®), ensuring the same safety, efficacy, and clinical outcomes. |
| cGMP Manufacturing Standards |
Manufactured in strict accordance with FDA regulations and Current Good Manufacturing Practices (cGMP), guaranteeing consistent product quality and purity. |