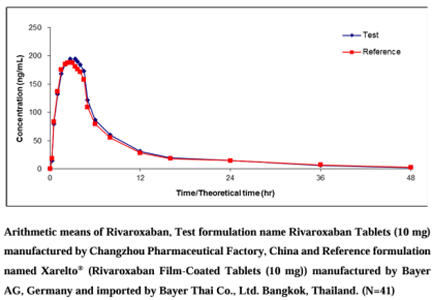
Rivaroxaban Tablets
Strength: 10 mg, 15 mg, 20 mg
1.Product Overview
| Product Name | Rivaroxaban Tablets, USP |
|---|---|
| Reference Listed Drug (RLD) | Xarelto®(Janssen) |
| Pharmaceutical Form | Film-coated tablet |
| Strength | 10 mg, 15 mg, 20 mg |
2.Indications and Usage
Rivaroxaban is a Factor Xa (FXa) inhibitor indicated for:
| Nonvalvular Atrial Fibrillation (NVAF) | Reduction of the risk of stroke and systemic embolism in adult patients |
|---|---|
| Treatment of Deep Vein Thrombosis (DVT) | Treatment of DVT in adult patients |
| Treatment of Pulmonary Embolism (PE) | Treatment of PE in adult patients. |
| Reduction in Risk of Recurrence of DVT and/or PE | In adult patients at continued risk for recurrent DVT and/or PE. |
| Prophylaxis of DVT (Post-Surgery) | Prophylaxis of DVT in adult patients following hip replacement or knee replacement surgery. |
| Chronic Coronary Artery Disease (CAD) / Peripheral Artery Disease (PAD) | Reduction of the risk of major cardiovascular events (CV death, Myocardial Infarction, and stroke) in adult patients. |
3.Dosage and Administration
Important Administration Instructions:
| 15 mg and 20 mg tablets | Must be taken with food (specifically, with the evening meal for NVAF). |
| 10 mg and 2.5 mg tablets | Can be taken with or without food. |
Dosage adjustments are required for patients with renal impairment (based on CrCl).
Standard Adult Dosing:
| Indication | Dosage | Duration |
|---|---|---|
| Nonvalvular Atrial Fibrillation (NVAF) | 20 mg once daily (with evening meal) | Long-term |
| Treatment of DVT / PE | Initial: 15 mg twice daily (with food) | 21 days As indicated |
| Maintenance: 20 mg once daily (with food) | As indicated | |
| Prophylaxis of DVT (Hip/Knee) | 10 mg once daily | Varies (e.g., 35 days post-hip, 12 days post-knee) |
| CAD / PAD | 2.5 mg twice daily (in combination with low-dose aspirin) | Long-term |
4.Comparison of Direct Oral Anticoagulants (DOACs)
| Feature | Rivaroxaban | Apixaban | Dabigatran | Edoxaban |
|---|---|---|---|---|
| Mechanism of Action | Direct Factor Xa Inhibitor | Direct Factor Xa Inhibitor | Direct Thrombin (IIa) Inhibitor | Direct Factor Xa Inhibitor |
| Standard Dosing (for NVAF) | 20 mg Once Daily | 5 mg Twice Daily | 150 mg Twice Daily | 60 mg Once Daily |
| Food Requirement (High Dose) | Must be taken with food (15 mg & 20 mg tablets) | No food requirement. | No food requirement (capsule must not be opened/crushed). | No food requirement. |
| Key Differentiator | Once-daily dosing for NVAF and VTE maintenance. Requires food for high-dose absorption. | Twice-daily dosing. Considered by some studies to have a favorable GI bleeding profile. | Twice-daily dosing. The only direct thrombin inhibitor in oral form. | Once-daily dosing. Requires 5-10 days of parenteral (e.g., heparin) lead-in for acute VTE treatment. |
5.Advantages of SPH Rivaroxaban Tablets
| Cost-Effectiveness | Significantly reduces the treatment cost compared to the brand-name drug (Xarelto®), making long-term anticoagulation therapy more accessible and sustainable for patients and healthcare systems. |
|---|---|
| Therapeutic Equivalence (A-Rated) | As an FDA-approved generic (ANDA 216995), it is bioequivalent to Xarelto®, guaranteeing the same high standards of safety, efficacy, and pharmacological performance. |
| cGMP Manufacturing Standards | Manufactured in strict accordance with the FDA's Current Good Manufacturing Practices (cGMP), ensuring consistent product quality, purity, and potency. |